|
Noble gases can also be used to produce excimer lasers, especially in combination with halogens. Xenon produces a nearly continuous spectrum that mimics sunlight well, so it is a popular choice for solar simulators. Bulbs filled with neon for example will produce light with an orange-red colour, but you can change this emission with the use of phosphor additives. The choice of noble gas used can change the colour of light that can be produced. By surrounding light sources with noble gas, manufacturers can reduce the rate at which the filament burns, increasing lamp lifetime. Noble gases are often used as a filler gas within arc or filament lamps. Also, for applications such as welding, nitrogen is not a great choice as it will become reactive in the presence of an electric arc. If you are working with materials that can react with nitrogen (such as lithium, magnesium, and calcium carbide) you may wish to use Argon gas. 
Additionally, argon is a noble gas, unlike nitrogen. There are situations where it may be worth the extra money though, for example if you are working with very sensitive materials. However, it is significantly more expensive than using nitrogen gas as argon is much less abundant. It will liquify at -186 ℃, so argon gas is cheaper than helium to store. It is heavier than both nitrogen and helium, so will be the most effective inert gas at displacing air within a confined space. Argon is also used in combination with nitrogen in filament bulbs as it decreases burn out rate of the filament.Īrgon is the most popular noble gas used in glove boxes. Additionally, you can use solid argon to trap extremely air sensitive material within a solid matrix in order to study them. By filling a contained space with argon, air-sensitive compounds can be handled without reacting with the environment. Argon electron configurationĪrgon is most commonly used to process air sensitive materials. It is the 3 rd most abundant gas on earth and is much heavier than helium. ArgonĪrgon has an atomic number of 18 and has an outer shell containing eight electrons. For example, helium glove boxes are sometimes used to conduct studies involving lithium batteries. However, there are some materials which require this level of inertness - such as lithium. However, due to the low abundance of helium on earth and the high temperatures/high pressures needed to store liquid helium, it is an expensive choice - especially for high purity helium. The stability of helium makes it a good material to conduct air sensitive experiments in. Therefore, these superconductors can be used to create powerful magnets that can used in nuclear magnetic resonance (NMR) imaging. Submerging these materials in liquid helium basically eliminates all electrical resistance. One use of liquid helium is to cool certain materials enough so that they exhibit superconductivity. 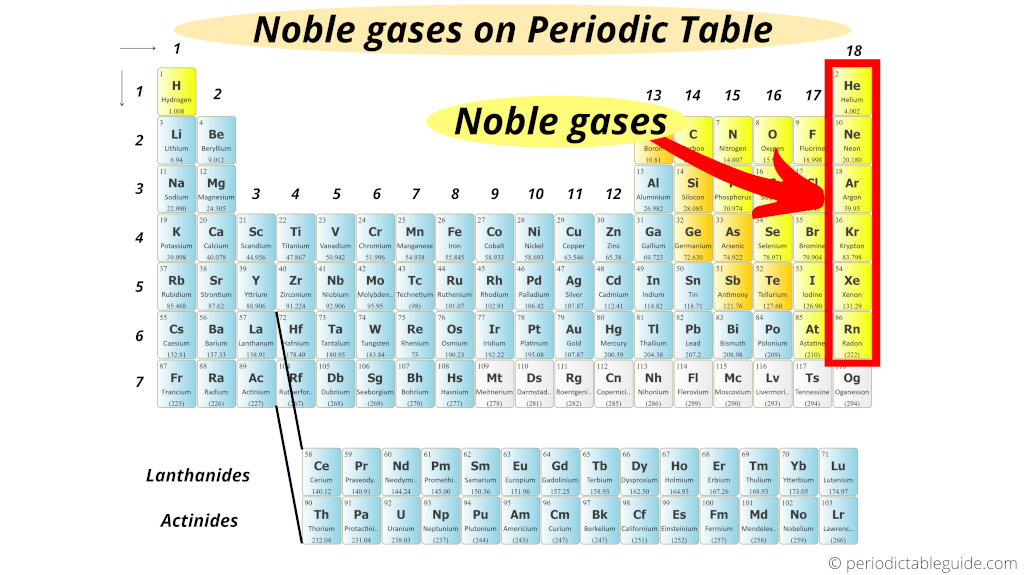
Helium liquifies at 4.18 K (or -268.97 ℃) so storage of liquid helium requires extremely low temperatures or high pressures. Upon being released into the air, it will quickly escape terrestrial atmosphere into space. Helium is a lot less dense than most of the elements in the earth's atmosphere. It is one of the most abundant gases in the universe, but only the 71 st most abundant element on earth, with most of it being found under the Earth's crust. This is a noble gas so it can be used to create an inert environment. Helium is the simplest noble gas, holding only two electrons in one electronic orbital. 
We have discussed helium and argon, as these are primarily used for their inert properties. Noble gases are therefore optimum choices to use for inert processing conditions. This means they will not take part in chemical reactions, and it takes a large energy input to create molecules using these atoms. These gases have full outer orbital layers, so they neither want to take electrons from or donate electrons to other atoms. These all exist as gases at standard temperature and pressure. The term noble gas refers to atoms that are in group 18 of the periodic table. Other atoms can form stable compounds by forming bonds with like atoms, such as nitrogen. Noble gas atoms have full outer electrons inherently. This means they have no free electrons or vacancies to interact with other molecules. Inert atoms or molecules have full outer orbitals. The reactivity of an atom depends on its electronic configuration. However, nitrogen gas (N 2) is also very unreactive so is also considered an inert gas in many fields. The term inert gas is often used interchangeably with the term "noble" gases (helium, argon, xenon) - which refers to atoms in Group 18 of the periodic table.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
